What does GxP stand for?
GxP stands for Good X Practice (FDA compliance; X can mean: Clinical, Distribution, Laboratory, Manufacturing, Cultivation, Pharmaceutical, etc.)
Med-Prov, LLC uses GxP Standard Practice coupled with Patient Safety Standards to assure regulatory bodies, patients and customers will trust Cannabis growers and Cannabis products manufacturers and distributors to provide cannabis free from heavy metal. We use Certified Regulatory Professionals to provide Good Standard Practice solutions for both Government and Private Organizations through Deviation and Risk Management controls holding our customers to higher GXP Compliant standards.
RELY ON MED-PROV FOR ADVOCATING AND SUPPORTING YOUR PRODUCT SAFETY AND INTEGRITY.
We do this by collaborating with organizations to establish and design Policies and Protocols founded on the precepts of Good Distribution Practice (GDP), Good Storage Practice (GSP), Good Manufacturing Practice (GMP) and Good Clinical Practice (GCP). Our associates have extensive knowledge of GXP and Quality Management standards.
GMP Validation and Compliance Requirements:
We offer the highest quality of Regulatory Audit Services in addition to Quality Validation for Pharmaceutical, Medical Device and Diagnostics, Facility and Utilities, Equipment, Methods and Computer Validation. Meed-Prov is committed to providing you with exceptional validation and testing services and staying current with industry best practices, amended standards and continued trends.
GXP Consultants are committed to providing our clients with a superior level of validation consulting and testing services and keeping them current with validation issues, standards and trends. GXP Consultants dedicated validation laboratories provide you with your validation testing and methods development needs.
Compliance solution for your needs include:
- Validation Master Planning and Gap Analysis
- Validation Project Management
Med-Prov is prepared to help you with the development and/or review of functional requirements, system design review, system specifications, development of standard operating procedures and preventive maintenance records and vendor evaluations and audits. Our team will provide you with comprehensive support for Complied Testing, Analytical Method Development and Validation for Medical Devices and Pharmaceuticals.
- Current Compliance Services using good practice rules (GxP):
Compliance Program Design and Evolution - Internal/External Audits of Company Practices
- CIAs, IROs and Key Negotiation Points
- Extensive review of Good Practice (GxP) and Quality Management standards
- Development of a company-wide GxP policy
- Successful implementation of additional processes that enhance the company’s current ISO 9001:2008 program
- A corrective and preventive actions (CAPA) program
- A change management program
- A management review program\
- A process performance/product quality monitoring program
- Standard operating procedures, as well as global and local instructions
- An internal audit program (QA)
- A self-inspection program (QC)
- Complaint management
- A risk assessment approach for decision-making
- Qualification/validation programs
- Calibration/preventive maintenance program
Deviation Handling: Our team uses best-practices and Regulations developed by the World Health Organization’s Quality Risk Management Approach:
Event Detection
- Deviation Categorization
- Root Cause Investigation
- Corrective and Preventive Actions (CAPA)
- Quality Risk Management
Quality Risk Management Steps
- Risk Assessment
- Risk Control
- Risk Review
- Risk Communication
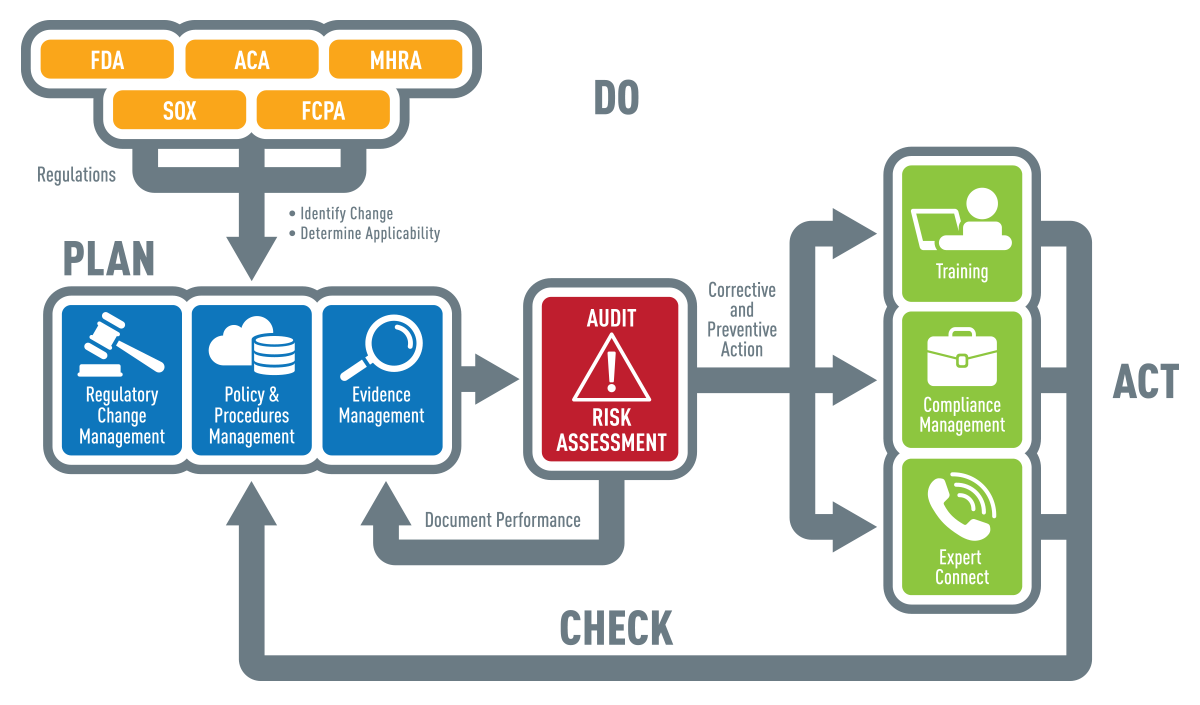
FDA GXP Compliance Management Process
4 Types of Cannabis Operations with differing Requirements
Med-Prov is prepared to support your operation in meeting legislative and regulatory requirements. We base a large part of our training and knowledge from the National Cannabis Standards coupled with partnering with a reputable association whose primary focus is patient focused and patient safety.
The National Cannabis Standards (NCS) guide operators to maintain compliance with the National Cannabis Standards as well as local and state based standards. Med-Prov uses these standards and other resources to address issues related to the safe use and responsible commerce of legally marketed products derived from Cannabis species. These NCS guidelines are applicable to cannabis and hemp as well as cannabis-derived and hemp-derived products.
Cultivation Operations:
 It’s extremely important that you understand your operation so you may implement relevant mandates. Cannabis may be cultivated in a number of facility settings including indoors, in a greenhouse, Outdoors as a field crop, and in nursery operations as allowed by applicable legislation and regulations. Cultivation operations often produce their own planting material and, in some Cases, may be allowed to obtain planting materials from other cultivation operations, nursery operations and compliant individuals.
It’s extremely important that you understand your operation so you may implement relevant mandates. Cannabis may be cultivated in a number of facility settings including indoors, in a greenhouse, Outdoors as a field crop, and in nursery operations as allowed by applicable legislation and regulations. Cultivation operations often produce their own planting material and, in some Cases, may be allowed to obtain planting materials from other cultivation operations, nursery operations and compliant individuals.
Processing operations most often obtain unprocessed plant material from cultivation operations and depending on applicable legislation and regulation may also obtain post-harvest plant material from other processing operations, from compliant individuals and from vendors. Most cultivation operations and or processing operations will primarily distribute raw plant material to dispensing operations and or manufacturing operations. In some cases, cultivation operations and processing operations may also be allowed to distribute cannabis to other cultivation operations, other processing operation, vendors, and direct Operations
Manufacturing, Packaging, Labeling, and Holding Operations
 Med-Prov’s Approach and Compliance Standards for Manufacturing, Packaging, Labeling and Holding operations may be applied to any person, group of persons, non-profit entity, or business entity that is engaged in the manufacturing, packaging, labeling, or holding operations for plant material or hemp, or plant material- or hemp-derived products.
Med-Prov’s Approach and Compliance Standards for Manufacturing, Packaging, Labeling and Holding operations may be applied to any person, group of persons, non-profit entity, or business entity that is engaged in the manufacturing, packaging, labeling, or holding operations for plant material or hemp, or plant material- or hemp-derived products.
However, a compliant individual that manufactures, packs, labels, or holds plant material, hemp, or plant material-, hemp-derived products in accordance with local and state law, solely for personal use or for another compliant individual and who does not engage in sales, is not subject to these standards.
While cultivation operations, processing operations and dispensing operations are not subject to these standards, this exemption does not apply to any off-site warehouse or storage facility that serves the cultivation, processing, or dispensing operation. In order to maintain compliance with the NCS for Manufacturing,.
Laboratory Operations
 Med-Prov’s Compliance solutions for laboratory operations may be applied to any person, group of persons, or business Entity that conducts analytical testing of cannabis, cannabis-derived Products, hemp, or hemp-derived Products.
Med-Prov’s Compliance solutions for laboratory operations may be applied to any person, group of persons, or business Entity that conducts analytical testing of cannabis, cannabis-derived Products, hemp, or hemp-derived Products.
However, a compliant Individual that conducts analytical testing of cannabis, cannabis-derived products, hemp, or hemp-derived products in accordance with local and state law, solely for personal use or for another compliant individual and who does not engage in sales, is not subject to these standards.
In order to maintain compliance with the National and Local Standards for laboratory, each operation is responsible to comply with only those provisions of the legislation that apply to the activities conducted by the operation.
Dispensary Operations
 Dispensing operations must be in compliance with all legal requirements in the jurisdiction where the dispensing is occurring. Dispensaries MUST implement effective best practices including relevant Policies and Procedures. We can assist your Dispensary meet all regulatory and legal requirements to minimize risks and liabilities.
Dispensing operations must be in compliance with all legal requirements in the jurisdiction where the dispensing is occurring. Dispensaries MUST implement effective best practices including relevant Policies and Procedures. We can assist your Dispensary meet all regulatory and legal requirements to minimize risks and liabilities.
Types of dispensing operations could include:







